Vesicoamniotic Shunting For Lower Urinary Tract Obstruction: Renal Parenchyma Area On Prenatal Ultrasound as a Predictor Of Kidney Function.
Paulo R. Moscardi, M.D.1, Chryso P. Katsoufis, M.D.2, Marissa J. Defreitas, M.D.1, Rubén Blachman-Braun, M.D.1, Miguel Castellan, M.D.1, Andrew Labbie, M.D.1, Kristin Kozakowski, M.D.1, Rafael Gosalbez, M.D.1, Alireza Alam, M.D.1.
1Nicklaus Children's Hospital, Miami, FL, USA, 2Jackson Memorial Hospital, Miami, FL, USA.
Background: lower urinary tract obstruction (LUTO) in the fetus is associated with poor postnatal outcomes, including chronic and end stage kidney disease, and high mortality. Studies of the effect of fetal intervention through vesicoamniotic shunting (VAS) are marred by a device malfunction rate of up to 60%. Currently there are scarce biomarkers to prenatally evaluate and predict renal outcomes on these patients. Our aim was to assess the renal area measured by prenatal ultrasound (US) as a predictor of kidney function after birth.
Methods: this is a retrospective, cohort study of 16 male infants who survived the fetal intervention to birth. All had patent VAS in place at birth. Renal growth and size were measured using ImageJ Software (NIH) to calculate the total parenchymal area (TPA, defined as the area of the kidneys minus the area of the pelvicaliceal systems) in sequential prenatal US. These measurements were correlated with the kidney function status during follow-up. Demographics and clinical data were also recorded and analyzed.
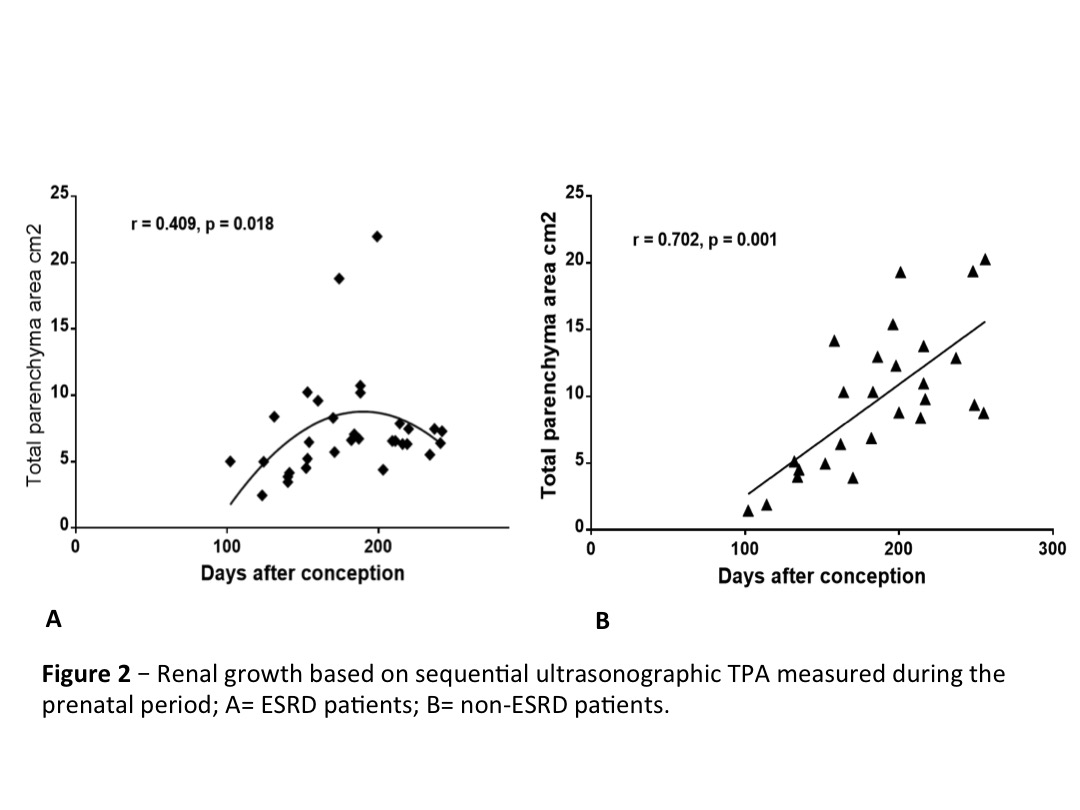
Results: Of the 16 males, 8 had posterior urethral valve, 4 had eagle-barret Syndrome, 3 had urethral atresia, and 1 was unknown. Shunts were placed at 21±3.5 weeks gestation and all neonates were born preterm (33.5±2.2 weeks) with low birth weight (2266±559 grams). Three patients died in the neonatal period, with 2 requiring renal replacement therapy (RRT). Mean follow-up of the remaining patients was 35.4±21.2 months. Fifteen patients had prenatal images available for analysis. We further divided the patients into 2 groups according to renal function status: 8 developed ESRD while 7 remained stable with CKD (non-ESRD). No difference was seen regarding the gestational age at prenatal diagnosis or VAS placement. Mean TPA during the second trimester was 5.8±4.0 and 5.0±2.4 cm2 for non-ESRD and ESRD respectively (p=0.56). These values were significantly different during the third trimester measuring with 11,9 ± 4,5 and 8,4 ± 4,26 cm2 for the non-ESRD and ESRD groups respectively (p<0.05) - figure 1. Moreover a strong correlation was observed in the two groups regarding the renal growth based on TPA measured on sequential prenatal US - figure 2. No difference was observed between groups in other prenatal US parameters, including: oligohydramnios, keyhole sign and bladder wall thickness.
Conclusions: even with definitive VAS for LUTO, postnatal morbidity and mortality remain high, emphasizing the role of renal dysplasia, in spite of urinary diversion, in postnatal kidney failure. TPA measurement during the prenatal period however, could have an important role as a non-invasive biomarker of postnatal kidney function.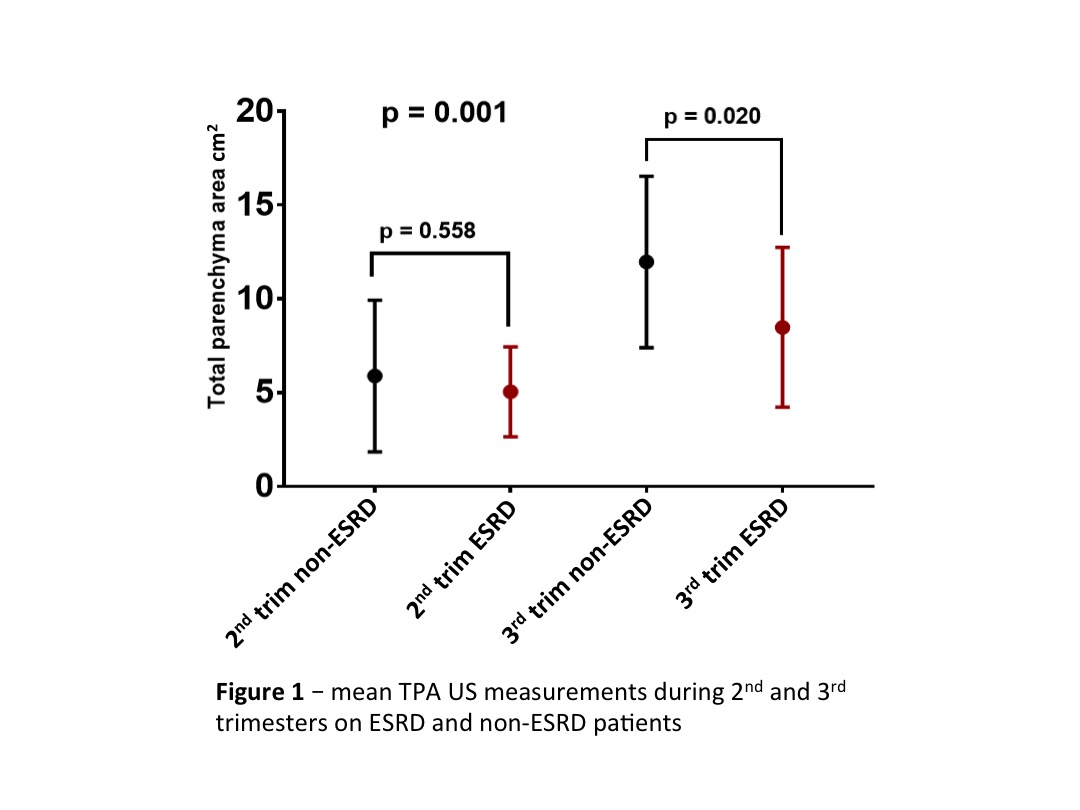
Back to 2017 Program
